24/07/2024
In 2017, the World Health Organisation estimated that between 35 – 40 million people require prosthetic and orthotic services. A growing and aging population, coupled with the rising incidence of diabetes and vascular related diseases contribute to an ever rising level of demand.
Although the common idea of a prosthetic is an artificial limb, the field of prosthetics covers the body in its entirety. Put simply, a prosthetic is an artificial device to replace or augment a missing or impaired part of the body. In broader terminology, a prosthesis is the name for the artificial body part itself.
A prosthesis may be visible to other people. Prosthetic limbs, prosthetic eyes, dentures and even wigs are examples of visible prostheses. Prostheses may also be internal to the body, for example: joint replacements, electronic voice boxes, and stents. External entities such as a dental brace, insoles or a pair of glasses are covered by the field of orthotics.
The Market
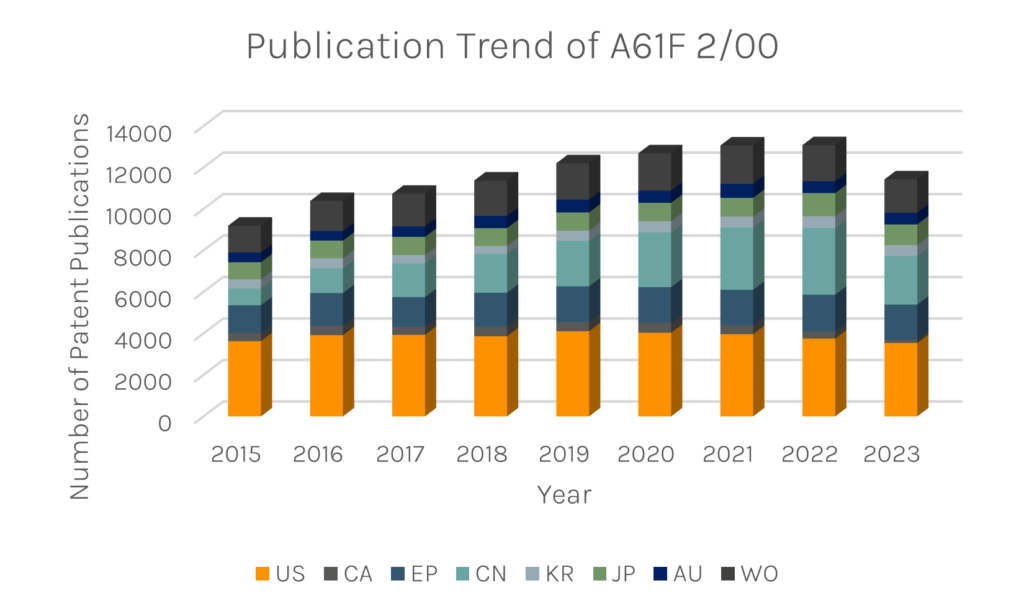
Taking a look at the worldwide patent publication trend for prostheses (IPC code: A61F 2/00), it is clear that there is a considerable commercial interest in the field. Companies are looking at the US and China in particular to protect their innovations. The vast number of publications each year gives an appreciation of the variety of prostheses that are being developed and improved.
A recent report by Polaris has valued the Prosthetics and Orthotics market at USD 6.76 billion in 2023, where it is anticipated to grow to USD 9.84 billion by 2032. Including the aforementioned reasons for the increase in demand, prostheses are becoming more widely known in developing economies, where the infrastructure for healthcare is improving. Use cases for prostheses are also extending beyond medicine. Prosthetics are becoming more popular in sports. Recent advancements in materials and technology has led to enhanced customisation, allowing a person to use a prosthetic that suits their particular body and needs.
Prostheses are becoming more sophisticated. Myoelectric prostheses, a myoelectric hand for example, closes the gap between a prosthetic and a real human hand. Myoelectric prostheses utilise sensors which detect electrical signals from a user’s residual muscles; these signals are converted into motion. Developments in materials science, manufacturing, neural interfaces and AI have all combined to make something like a myoelectric limb a reality. We will continue to see innovation in a plurality of scientific disciplines to further improve the level of sophistication of prostheses.
A key driver for further growth in the prostheses field is to sell the more advanced devices at more commercially accessible prices. This will open the door to a greater number of customers, particularly in developing economies. Another hurdle, particularly for prosthetics, is to continue to improve the design in an aesthetic sense. People will feel more accepting of the “new limb” if they feel self-confident wearing it.
Regulation
Regulation of medical devices is becoming increasingly stringent; the EU has introduced two new regulations on medical devices and in vitro medical devices, which came into force in 2021 and 2022 respectively.
The recent EU medical device regulations “significantly tighten the controls to ensure that medical devices are safe and effective and at the same time foster innovation and improve the competitiveness of the medical device sector.” Although the regulations have been brought about to incorporate the latest progress in science and innovation, they were necessitated by a PIP breast implant scandal and faulty metal-on-metal hip implants, which put the old system under scrutiny. Both of these cases involve prostheses; they illustrate the significance of the quality and reliability required in a prosthesis. It is also important to point out that the two cases, especially the PIP breast implant case, are comparatively low risk devices, say, in view of a pacemaker. However, companies should not have the opportunity to cut corners simply because a prosthesis isn’t in a high-risk category. This is a major reason why the EU has implemented these regulations. It will be interesting to see what effect regulations will have on the prostheses field in the future, especially with regard to lower risk prostheses.
Ongoing regulation to enhance the safety of medical devices, including prostheses, will occur around the world. These regulations may impose more stringent rules in the development process. However, this cannot be conflated with stifling innovation in the sector. The safety of the end user is paramount. Companies will adapt accordingly to create and improve upon new medical devices like prostheses. We will continue to see growth in the sector as demand continues to rise and innovation opens doors to new prostheses, so we hope that this demand is met with reliable and safe prostheses.
Here at Reddie & Grose, we have a large number of attorneys in our Medical Devices team with interest and experience in such technologies. If you need help protecting your innovations in this field, then please do get in touch.
This article is for general information only. Its content is not a statement of the law on any subject and does not constitute advice. Please contact Reddie & Grose LLP for advice before taking any action in reliance on it.



